  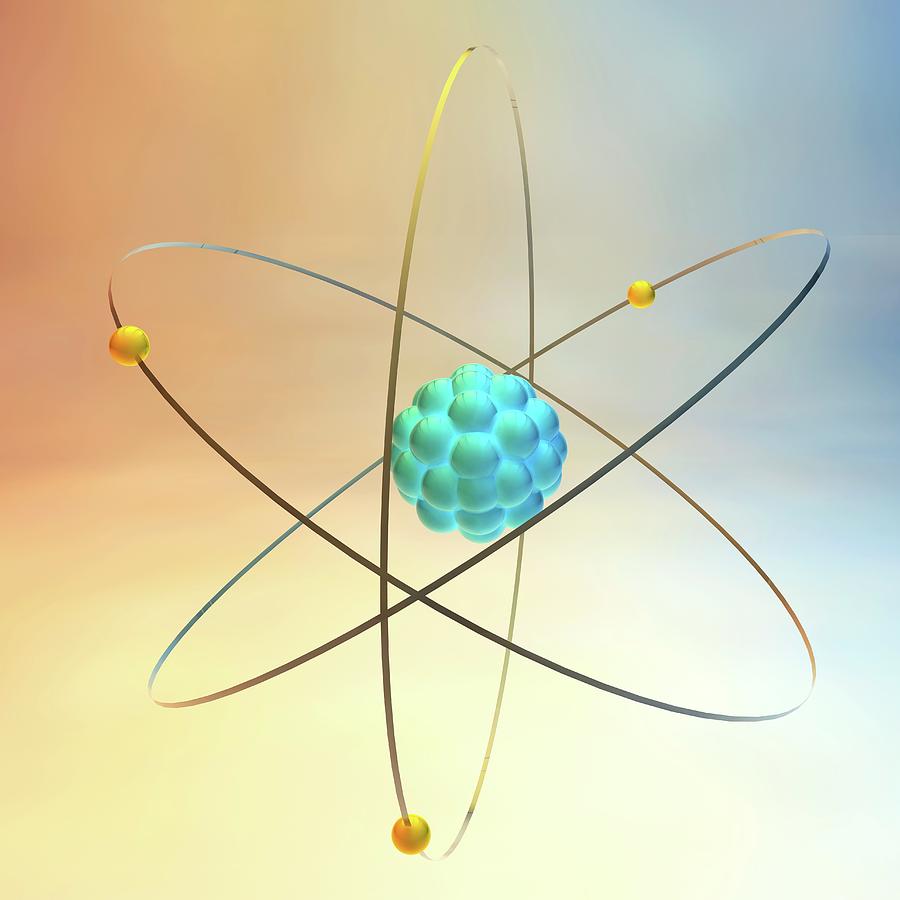
When referring to nuclides of the same element they are generally term isotopes i.e. Nuclides are often identified using the name of the element and the mass number (total number of protons and neutrons), i.e. Elements with atomic numbers 1 – 98 have all been shown to exist in nature while elements with atomic number 99 – 118 have only ever been produced artificially. As an example hydrogen has three (3) isotopes: hydrogen-1 (hydrogen), hydrogen-2 (deuterium) and hydrogen-3 (tritium) (Figure 3).Īt present there are 118 known elements which are typically displayed on the periodic table of the elements. The mass of the nucleus in atomic mass units (amu) is usually slightly different from the mass number.Ītoms of the same element can have different number of neutrons and they are called isotopes of that element. The mass number (A) of the nucleus is equal to Z + N (Figure 2). The number of neutrons in the nucleus is denoted by N. The number of protons in the nucleus is called the atomic number (Z), the atomic number defines the element. This force is much stronger than the electrostatic force that binds electrons to the nucleus, but its range is limited to distances of the order of 1 x 10 -15 meters. See Figure 1. Nuclei are made of positively charged protons and electrically neutral neutrons held together by a nuclear force. An atom is the smallest unit of an element that will behave as that element.Ītoms consist of an extremely small, positively charged nucleus surrounded by a cloud of negatively charged electrons. Although typically the nucleus is less than one ten-thousandth the size of the atom, the nucleus contains more that 99.9% of the mass of the atom. oxygen, carbon, uranium) are made up of different types of atoms. 
Atoms are the extremely small particles that are the basic building blocks of ordinary matter.Ītoms can join together to form molecules, which make up most objects. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
